1 2 3 4
A (H-1A (U-4A) (M-Cu2B) (H-V3B)
B (M-1A) (H-Cu1A) (U-Cu2B) (M-V3B)
C (U-1A) (M-Cu1A) (H-V2A) (U-V3B)
D (H-3A) (U-Cu1A) (M-V2A) H2O
E (M-3A) (H-Cu4A) (U-V2A) H2O
F (U-3A) (M-Cu4A) (H-V3A) H2O
G (H-4A) (U-Cu4A) (M-V3A)
H (M-4A) (H-Cu2B) (U-V3A)
Above are for Dec 7, 2010 run. For December 2, 2010 run, H-U-V3B in D4-F4, H2O in A4-C4
The spreadsheet was public, so anybody should have been able to view or edit it. Maybe it's a compatibility issue between UW and Gmail. But I've since made it private and tried adding everybody in the oyster group privately. Hopefully, it works. -
I don't have permission to edit the google doc's spreadsheet, whoever is in charge of that can you add me so that I can add my western blot data? -
Try this link -
It still says prattp@uw doesn't have access to the spreadsheet-
December 8, 2010
Link to Oyster qPCR Analysis -
Reformat -
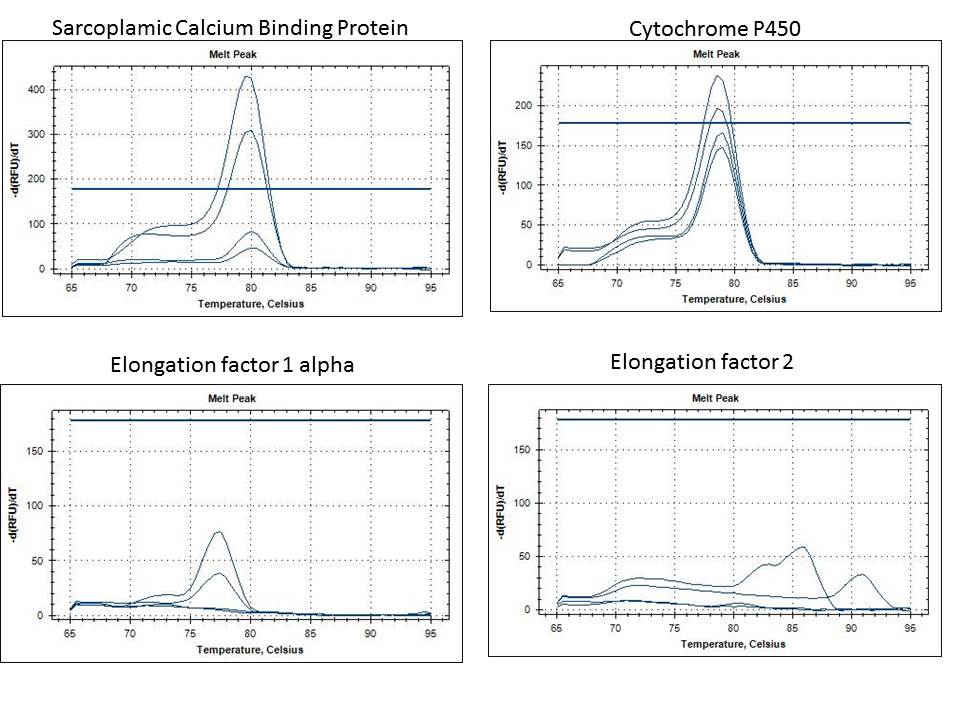
needs to be on single sheet, I started the sheet and did 18s
All oyster gene expression data including Ross's methylation data should be added to this sheet. For Ross, raw Cts are fine.
December 6, 2010
Link to most recent Draft of Oyster paper: Oyster Draft
-
December 2, 2010
RNA concentrations were normalized again for a final end concentration of 2 ug using the following equations:
1. (RNA conc ng/uL)/1000= RNA conc ug/uL
2. (RNA conc ug/uL) * X = 2 ug ---> X = 2 ug/(RNA conc ug/uL) where X equals the amount of stock RNA to add to the reverse transcription for RNA
The following google doc is the data and calculations for each sample: https://spreadsheets.google.com/ccc?key=t7fhoi_PCVo_3vcUnIIPQAA&hl=en#gid=0
*Note because final RNA amounts vary for each sample the amount of nuclease free H2O will vary as well and was calculated by water=(5-RNA)+4 and these calculations are included int the above spreadsheet as well*
All samples were reverse transcribed using the following protocol:
- Thaw and mix RNA stock tube by inverting
- Add pre-calculated stock RNA, 1 μl oligo dT, and individual amount of nuclease free H20 based off of RNA amount added in a 0.5 ml PCR tubes
- Return RNA stock sample to ice
- Incubate mixture for 5 min at 70°C on the thermocycler then put tube on ice till cool.
- Briefly centrifuge sample tube
- Add 5 μl of M-MLV 5X Reaction Buffer, 5 ul of dNTPs, 1 μl of M-MLV RT, 4 μl of nuclease free H2O to sample tube.
- Incubate mixture for 60 minutes at 42°C and then heat inactivate at 70°C for 3 minutes on the thermocycler.
After reverse transcription each sample was transferred to its own individual labeled 1.5 mL microcentrifuge tube and an additional 225 uL of nuclease free water was added for a final dilution so enough product is available for qPCR's of three different genes in duplicates
All samples were returned to the -20°C for storage
-
November 30, 2010-December 1, 2010
RNA was extracted from oyster gill tissue from Big Beef Creek. RNA concentrations were recorded on a Nanodrop Spectrophotometer. Most of our RNA concentrations were at or around 3000 ng/µL. From those initial concentrations, using (C1)(V1)=(C2)(V2), we calculated how much concentrated RNA is needed to make a 500 µL of 200 ng/µL RNA stock. The math was checked multiple times, but after making dilutions of Control 1A and 2A, we again measured RNA concentration. We got readings of 20 ng/µL and 40 ng/µL, respectively. We didn't want to lose any more time by continuing to make dilutions. After talking to Steven and Caroline, we decided to DNase 50 µL of our concentrated RNA extract and then quantify our RNA concentrations again. Turbo Free DNase kit/protocol was used. The concentrations are in the range of 500-1800 ng/µL. Currently, they are labeled and sitting in the -80 C freezer. We need to thaw them, centrifuge them, extract the liquid (which should now be purely RNA suspended in 0.1% DEPC). We can run qPCR with this RNA stock. In order to do so, we need between 1 and 10 µg of RNA. We need to calculate how many µL of stock we need to get between 1 and 10 µg of RNA.
The next step is to extract only the suspended RNA and to leave behind the broken down DNA at the bottom of the tubes. A lot of 4B Vt sample evaporated during the DNase incubations.
Here are the concentrations
| Sample Number |
Control [RNA] (ng/µL) |
Cu [RNA] (ng/µL) |
Vt [RNA] (ng/µL) |
Cu+Vt [RNA] (ng/µL) |
| 1 A |
769.9 |
812.7 |
1479.1 |
1226.0 |
| 2 A |
1355.2 |
1304.7 |
1297.8 |
1403.1 |
| 3 A |
930.7 |
1319.8 |
1649.6 |
1382.1 |
| 4 A |
817.0 |
1764.3 |
1387.0 |
(no sample) |
| 1 B |
1119.8 |
940.8 |
863.1 |
885.5 |
| 2 B |
594.2 |
1567.1 |
1454.5 |
1052.8 |
| 3 B |
1514.8 |
1941.4 |
2214.9 |
1529.2 |
| 4 B |
(no sample) |
1524.1 |
1025.8 (not much sample is left but there's plenty of RNA in what we have) |
1030.1 |
Our 260/280 was good, falling in a range between 1.88-2.02... Most were right around 1.91.-
Unless you have already signed-up, the qPCR thermocyler is full for this week. -
Also, we will be scaling down our qPCR reactions to a volume of 25ul. That is:
For a 25μl reaction volume:
| Component |
Volume |
Final Conc. |
| Master Mix, 2X (Immomix) |
12.5µL |
1x |
| Syto-13 dye (50uM) |
1µL |
2µM |
| upstream primer, 10μM |
1.25μl |
2.5μM |
| downstream primer, 10μM |
1.25μl |
2.5μM |
| Ultra Pure Water |
7uL |
NA |
Molecular projects
Dave M. - I will be looking at changes in V. tubiashii gene expression under the different experimental conditions of seawater, seawater + Cu, seawater + oyster, and seawater + oyster + cu. I will need to extract RNA from the water filter samples collected on 11/19/10
Ross - Looking at methylation at five sites as well as globally. Need to extract DNA from the gill tissue we collected.
Paul- Western Blot
Jason - I'm looking at Metallothionein IV gene expression in oyster gill tissue, which is full of hemocytes.
Sammi - I will be looking at HSP 70 gene expression in oyster gill tissue
November 19, 2010
Tissue Sampling
*Can someone expand on this that was there the whole time? I was running around a lot and dont have a grasp on the details... -
Today the group is collecting oyster tissues from the Cu/Vt challenge that has been going on all week. Tissues to be collected are the gill (for its concentration of hemocytes), mantle (mostly for DNA/methylation analysis), hemolymph (alternative source for hemocytes), and size dimensions (Lenth, width, height) of the oysters.
Tasks
Oyster Shucker - split between Jason and Ross
Gill tissue - Sammi
Hemolymph - David
Mantle - Paul
Size measurements - Dave and Jason
Water filtering - Dave
November 18, 2010-
V.tubiashii challenge
Vt culture from 11/17/10 was spun down at 4,800rpm for 20min in a swing bucket rotor.
Remove supt. and resuspend Vt in 1L of filtered seawater.
Spin again 4,800rpm 20min.
Remove supt. and resuspended in 110ml filtered seawater (11 treatments, 10ml/treatment)
Spec at OD550.
Spec Reading = 0.750
Calculating CFU
Estimated CFU by assuing 1U at OD550 = 5x10^8 cfu/ml
0.750 x (5x10^8) = 3.75x10^8cfu/ml
3.75x10^8cfu/ml x 10ml/sample = 3.75x10^9cfu
3.75x10^9cfu = total amount of Vt added to each bucket
3.75x10^9cfu/5000ml = 7.5x10^5
7.5x10^5cfu/ml = concentration of Vt in each bucket
DISPOSAL of V. tubiashii exposed oysters: Discard tissue and shell waste in autoclavable biohazard bags which are in the lab. After sampling, get Caroline. I will take the waste to FSH to be autoclaved. Once the exposed oysters have been autoclaved I will put them in MER -20C walk-in for KH to dispose of. -
ATTENTION!!
While updating the methods section of the google doc I noticed the math for the ppm calculations of Copper sulfate were off 100 fold. Instead of adding the "EPA relevant" 1.3ppm, 130ppm was added. We need to do some research ASAP on what this means, is it ecologically relevant? Are there places with Cu concentrations this high? Fortunately we will still have the vibrio challenge, but we need to determine as quickly as possible if the copper treatment will generate meaningful data... -
| SammiBrombacker |
I checked the calculations we did and I'm still getting 6.5 mg to 5 liters comes out to 1.3 ppm I think i must be doing it wrong then what are the correct calculations-
As of 2:30pm 11/17/10, the oysters are still alive... -
Cu ppm findings
352mgCu/kg sediment dry weight found in duwamish during 2007 survey. -
Cu ranges can be up to 300 ppm in soil when hydrous iron and manganese oxides --http://www.springerlink.com/content/u236126j77735k74/fulltext.pdf
C. gigas Cu uptake will lead them to turn green and were recorded to have an daily acclimation rate of 216 ppm/Cu --http://www.sciencedirect.com.offcampus.lib.washington.edu/science?_ob=MImg&_imagekey=B6VB5-4903NJS-G-1&_cdi=5917&_user=582538&_pii=026974919090126W&_origin=search&_coverDate=12/31/1990&_sk=999349995&view=c&wchp=dGLzVzb-zSkzV&md5=e2db3fd1483448aa3b8c5e980e9b7e82&ie=/sdarticle.pdf
When looking at sediments from clean and dirty sites this paper found varying effects at different ppm's of numerous heavy metals in the sampled sediment, Cu included, and researched effects on embryogenesis in C. gigas fertilized eggs. They saw toxic effects on the eggs at 500 g/L but no toxic effect in any concentration less than that --http://ql3dq7xx6q.search.serialssolutions.com.offcampus.lib.washington.edu/?&url_ver=Z39.88-2004&url_ctx_fmt=info:ofi/fmt:kev:mtx:ctx&rft_val_fmt=info:ofi/fmt:kev:mtx:journal&rft.atitle=Effects%20of%20storage%20temperature%20and%20duration%20on%20toxicity%20of%20sediments%20assessed%20by%20Crassostrea%20gigas%20oyster%20embryo%20bioassay&rft.auinit=R&rft.aulast=Beiras&rft.date=1998&rft.epage=2105&rft.genre=article&rft.issn=0730-7268&rft.issue=10&rft.spage=2100&rft.stitle=ENVIRON%20TOXICOL%20CHEM&rft.title=ENVIRONMENTAL%20TOXICOLOGY%20AND%20CHEMISTRY&rft.volume=17&rfr_id=info:sid/www.isinet.com:WoK:UA&rft.au=His,%20E&rft.au=Seaman,%20M
67% survival rate at 1.0 ppm exposer of C. gigas -- http://www.springerlink.com/content/x444l817h7728t42/fulltext.pdf
Found that some fresh water snails started to behave oddly above .36 mg/L and would produce excess mucous and release gametes -- http://www.extension.org/article/18351
Looking at a few abstracts (I can't read the papers w/o paying for it off goggle scholar) it seems that there can be extremely high Cu concentrations in estuary sediments from plant, mining, etc. runoffs that were rich in Cu and oysters were surviving in the areas -
| SammiBrombacker |
King County Department of Natural Resources and Park reported that stormwater from the SR 520 Bridge in Seattle contained copper at levels ranging from 36.0 – 76.5 μg/L -
| sr320 |
 |
| 0903051.pdf (page 28 of 51) |
Uploaded with Skitch !
November 17, 2010
Vt does not appear to be growing -
| sr320 |
Tending to the Vt cultures... -
After o/n incubation of 100mL cultures of Vt and Vp at 37degrees C, it looks like the Vt has not grown at all.
Started three new 5ml cultures of Vt
One culture was inoculated from a Vt plate into the LB (1%NaCl) that was prepared yesterday
Another from the same plate but into Marine Broth
and the third was from a -80 glycerol stock into the LB made yesterday.
NOTE: After innoculation, I added another 10g (1%) NaCl to the LB, split it into two 500ml aliquots, and reautoclaved.
At the end of the day today, I will take all of my Vt cultures and combine them into one of the 500ml flasks of LB, and add the Vp culture that is growing so well into the other 500mL aliquot of LB
One pair of Metallothionein IV primers ordered! -
Sampling wish list ceps YES
Oyster Shuckers YES (5)
labeled cryo viles.....check!
Liquid Nitrogen
gloves YES (in lab)
sharps container YES
syringes YES
needles YES
kimwipes YES (in lab)
computer....ross?
calipers YES (3 sets)
Dewer and long forceps YES
-80 space
-80 boxes...check!
Bleach, water, ethanol, and fire for sterilization of dissection tools.
__Molecular reagent/equipment wish list -__
| dmetzger1 |
TriReagent YES
EtOH (molecular grade) YES
DNA extraction kit
western reagents/electrophoresis boxes YES
dot blot reagents/equipment YES
qPCR master mix YES
DNase YES
Reverse Transcription reagents
more tubes and tips
ATTENTION GROUP! Please add to, change, or elaborate to anything on these lists ASAP!!!
November 16, 2010 -
| SammiBrombacker |
label tubes for friday -
| dmetzger1 |
| ddb4 |
6.5 mg copper/bucket for 5 L -
| SammiBrombacker |
| SammiBrombacker |
GSTO - hydrocarbon and pesticide response (from Ross) -- we only need to order Metallothionein IV gene (see below)-
| SammiBrombacker |
Accession number - AJ557141
Primer pair 3
| Sequence (5'->3') |
Strand on template |
Length |
Start |
Stop |
Tm |
GC% |
|||
| Forward primer |
ACAGGTTCCGCCTGTCCGGA |
Plus |
20 |
49 |
68 |
59.83 |
65.00% |
||
| Reverse primer |
ACCCGCCCCAGTGGGTTCTT |
Minus |
20 |
230 |
211 |
60.04 |
65.00% |
||
| Product length |
182 |
-
|
|||||||
LB was made and waiting to be autoclaved @ 3:30 pm LB WAS autoclaved at 4:30pm -
| dmetzger1 |
10 g NaCl
10 g Bactotryptone
5 g Yeast Extract
Mix on stir plate up to 800 ml with D/W
pH to 7.0
Add D/W to 1 L
Autoclave -
| SammiBrombacker |
Thursday @ 1:30 on 11/18/10 -- add Vt
Friday @ 1:30 on 11/19/10 -- sample
November 15, 2010 -
| jtayag |
Metallothionein IV gene, exons 1-4 for Crassostrea gigas is 2196 base pairs long (Accession number AM265551;http://www.ncbi.nlm.nih.gov/nuccore/AM265551.1 )
Primers designed using Primer3
| Primer |
Sequence |
Number of Bases |
% Guanine, Cytosine |
Melting Temperature |
| Forward |
taaaggcaaagccaagtgct |
20 |
45.00% |
60.02°C |
| Reverse |
ggtgcaccaaataagcgaat |
20 |
45.00% |
59.97°C |
I hate to be the bearer of bad news, but you picked the one sequence that is part of the promoter region which is not transcribed into RNA. I did a quick search on the NCBI site and there are a lot of exon/mRNA sequences -
| dmetzger1 |
Fixed primer design. Thanks. -
| jtayag |
November 12, 2010 -
| dmetzger1 |
Summary:
Oyster samples collected on November 11, 2010 put into system.
Talked with Sam and he started going V.tubiashii cultures on wednesday (11/10/10).
He also ordered 100g of CuSO4
Details:
There was a minor disaster this morning when I went to put the oysters into the basement system. Apparently the recirculating rates for the two sumps was not even which resulted in all of the water from one sump being pumped into the other. The problem is two fold:
1. One of the sumps was dry and not being held at 12 degrees.
2. The other sump was too full which lifted and tipped the buckets over.
All tipped buckets were drained and refilled with pure sea water. Half of the water from the full sump was pumped back to the dry pump. The chiller was disconnected from one of the sumps so it is now only circulating water for one system. I hooked up another chiller to the other system. This chiller is slighty different in that water is not flowing through it, instead there is a cooled wired that chills the water directly in the sump. This may or may not induce a tank effect. The pump was left in the water of the coil chiller to help mix the sump water.
Result: everything seems to be working for the moment and both sumps are holding at ~12-13 degrees C.
FINALLY the oysters can be put into buckets. Four oyster were put in each bucket. Blue/green label are from Big Beef Creek and Red labels are from the park. Treatments are separated so that all buckets that will eventually contain V.tubiashii will be kept together. Extra oysters were put in separate buckets and placed in the system with the coiled chiller.
No food has been added to the buckets.
Sammi will check on the systems before she leaves tonight.
November 11, 2010 -
| dmetzger1 |
Field Sampling!!
Low tide for the two sites were at 3pm. Low tides were a +7 which didnt allow us to collect oyster that were very far out on the tidal flats. Oysters at Big Beef Creak were collected closer to 4pm. ~40 oysters were collected from each site. Oysters were kept in coolers in the back of Steven's truck o/n and transported to the lab on friday morning.
November 10, 2010 -
| dmetzger1 |
Wet lab set-up for Oyster treatments:
With the help of Sammi and Ross we managed to hash out a system in the quarantine room in basement of FSH. Currently we have two large white totes (like the kind they use on fishing boats) that contain 8 buckets each. This gives us a total of 16 buckets which is how many we will need to carry out our 4 treatments in duplicate for each site.
At the moment we have seawater in all of the buckets with airstones only. There is no recirculation/filtration happening on the buckets. The waterbath totes have fresh water that is circulating through a chiller. Initial temperature measurements have one waterbath at 17 degrees C and the other at 19 degrees C. I will check the temps before I leave tonight to see if they are dropping towards our target temp of 12 degrees C.
(hopefully I can post a picture once we put animals in on friday)
Lose ends:
1. Will have to decide how we want to separate treatments. My vote is to keep all Vt treated animals in one tote and all control and copper only treated animals in the other.
2. Lids need to be made with labels for each bucket.
Photos from the field
 |
| Belfair |
Copper Treatment-
Decisions on that need to be made ASAP as will have to order. -
| sr320 |
Looks like the two forms most commonly used in the literature are copper sulphate and copper chloride.....copper sulphate looks to be more common. -
| dmetzger1 |
I also found a paper where Cu2+ was used -
| ddb4 |
Ordering info for Copper(II)Sulphate pentahydrate -
| dmetzger1 |
From this paper it looks like .5ppm would cause a response -
| dmetzger1 |
This paper used 10mg/l CuSO4
This paper has effects of copper at different levels
 |
| external image 20101112-n9gw8y336g2k8dtw2hq14irpag.png |
How to calculate ppm
(also indicates 1.3 ppm is EPA relavant)
Need to determine waste procedure for copper -
| sr320 |
Experimental Flow Chart: -
| dmetzger1 |
 |
| Slide1.jpg |
A few immune related papers of interest to me. -
| sr320 |
This paper is very similar to our study... -
| dmetzger1 |
Hey all! As per Steven's suggestion I have set the paper on google docs to 'private' in order to track changes made. This will require you to have a google account to log in. If you don't have one you can easily create one from the 'PAPER' link below. You will then have to get permission to edit the doc. There should be a link to contact me for that. If not, let me know and I'll add you manually.
-
| rosspitality |
Important links:
The Paper on Google Docs: PAPER
A Folder on Google Docs to Save References to: REFERENCES
Hello all! Here is our oyster stress project page. Enjoy!!!
-
| rosspitality |
